Optimising Clinical Development ways of working in a strategically outsourced model
4 min read 1 April 2025
A rapidly growing Global BioPharma organisation required our support to redesign and implement their clinical development process landscape and supporting ways of working. This followed their strategic decision to move from working with 10+ outsourcing providers with a variety of operating models, to a single strategic partner.
This shift represented a major change in legacy ways of working and roles & responsibilities for clinical development teams and functions. It was critical for the organisation to transform in a way that realised the business case of the strategic outsourcing partnership, remained compliant across all relevant aspects of GxP, and brought employees on an important change journey where they understood the enhanced value they could offer in this new model.
Redesigning 25 end-to-end Clinical Development processes in 9 months
Over a period of 9 months, we collaborated closely with the client team to fully re-design 25 key end-to-end clinical development processes across a range of diverse, cross-functional disciplines including Protocol Development, Biostatistics, Clinical Supplies, and Biosample Management; resulting in a 36% reduction in controlled documentation. Our approach involved broad engagement and co-design with cross-functional people to fully explore what their roles & responsibilities looked like in the new world, how to operate effectively with their strategic CRO partner, and what effective oversight looks like across a broad range of highly regulated activities.
Key to working across this breadth at pace was the rapid development and iteration of each future end-to-end process. This iteration was informed by the nature of the strategic CRO partnership and external best practices, including emerging regulations. It enabled business SMEs to quickly challenge and refine the processes, with a clear runway for further process development and go-live enabled by a highly engaged executive-level sounding board which was crucial for overcoming time-critical obstacles.
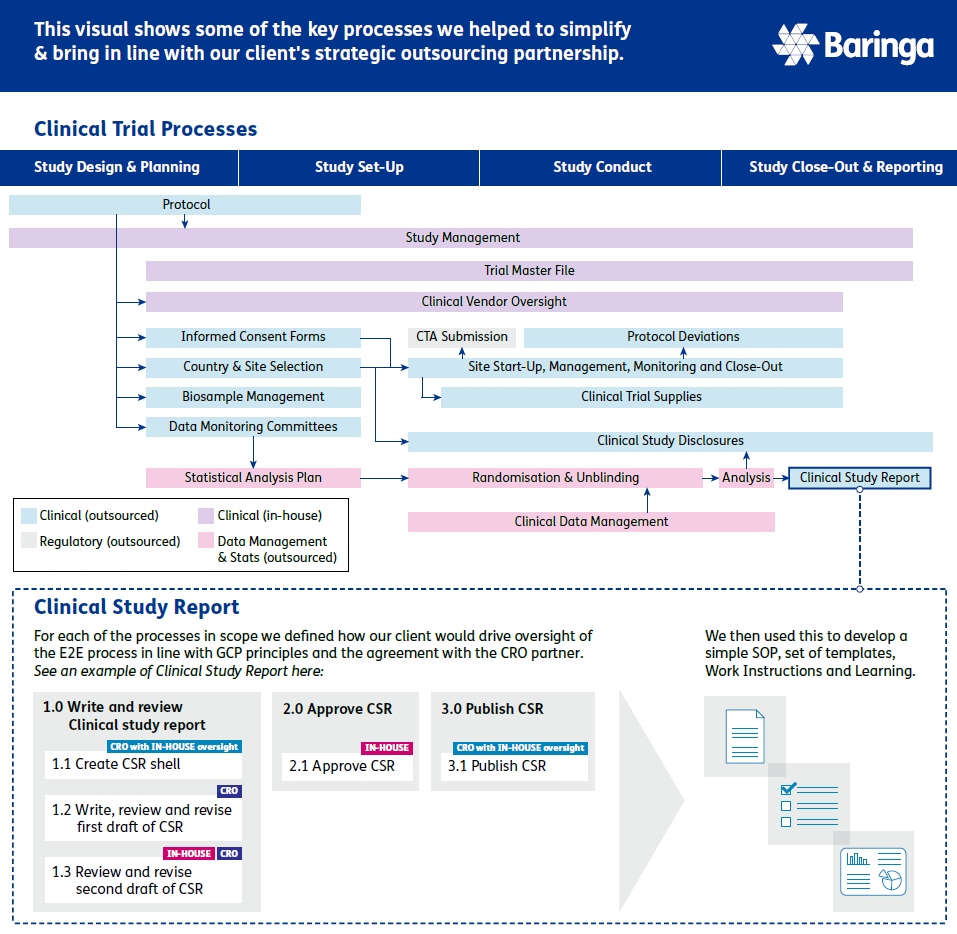
Creating an end-to-end, visual process architecture to strategically oversee and collaborate with the strategic CRO partner
Through this project, it was identified that the existing process landscape had been developed in functional siloes and that there wasn’t a full view of end-to-end clinical development (i.e. a process architecture) that everyone could understand. The benefits of developing such an architecture were clear to everyone: it would enable people to understand their role in, and work more efficiently across end-to-end activities; oversight of key activities would be easier to manage; and strategic oversight of the CRO partner would bet better enabled by understand the big picture and impacts.
An illustrated example of this process architecture can be found below.
This updated process architecture, with the ability to ‘heatmap’ oversight responsibilities and key risk areas relating to the strategic outsourcing partnership, has been integral to supporting the client with its ongoing, scaling clinical development goals.
Find out more about our work supporting powerful Partnerships in Pharma here or get in touch with Joao Pereira and Josh Elliott to discuss how we can help you.
Related Insights

The Power of Partnerships across the healthcare ecosystem: how can Biopharma take the lead?
The healthcare ecosystem encompasses a diverse range of organisations with a shared goal: to provide high quality and affordable healthcare. However, as this goal becomes increasingly challenging to maintain, Biopharma organisations play a key role in strategically partnering across the ecosystem to improve health outcomes.
Read more
Securing outsourcing success through strategic relationships with third parties
Discover how strategic third-party relationships in pharma outsourcing drive innovation, efficiency, and growth, transforming transactional partnerships into powerful alliances.
Read more
Unlocking the value of Digital and AI partnerships for Biopharma: in conversation with PathAI
Explore how digital and AI strategic partnerships for biopharma companies are shaping the future of precision medicine.
Read more
How powerful partnerships can unlock digital health innovation
Strategic partnerships offer an attractive route to unlock innovation within digital health however current failure rates sit at 60%. How can you build powerful partnerships?
Read moreRelated Client Stories

We talk to our client KCOM about modernising business operations
Join our TMT Director Sarah Roberts, as she talks to our client Tim Shaw, CEO at KCOM
Read more
Building a trusted data platform to enable scaling and cost savings
How do you accelerate delivery of a £multi-billion network expansion programme using advanced analytics and targeted insight?
Read more
Developing a Target State Operating Model for a technology and language services provider
Developing a comprehensive high-level target state operating model, defining the process architecture, and modelling the necessary capabilities for success.
Read more
Streamlining organisational structure to reduce complexity, scale, and grow
A new organisational resulted in a 10% cost efficiency across the organisation, as a result of simplification, standardisation, and the elimination of role duplication.
Read moreIs digital and AI delivering what your business needs?
Digital and AI can solve your toughest challenges and elevate your business performance. But success isn’t always straightforward. Where can you unlock opportunity? And what does it take to set the foundation for lasting success?